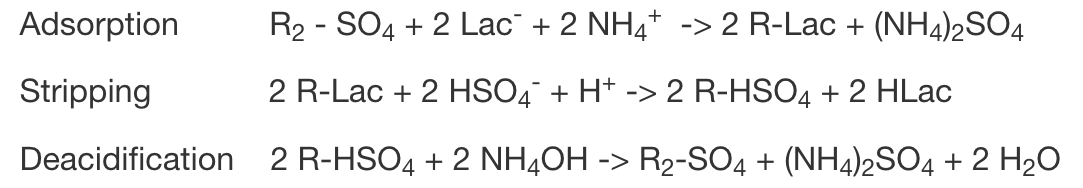Citric Acid/Lactic Acid
Citric Acid
Citric acid s a weak organic acid found in citrus fruits. It is a natural preservative and is also used to add an acidic ( sour ) taste to foods and soft drinks.
In biochemistry, it is important as an intermediate in the citric acid cycle and therefore occurs in the metabolism of almost all living things.
It also serves as an environmentally benign cleaning agent and acts as an antioxidant.
Many of the industrial applications of citric acid are based not only upon its acidic properties, but also upon its powerful sequestering action with various transient metals, such as iron, copper, nickel, cobalt, chromium and manganese.
Citric acid production by fermentation of molasses is still the major industrial route to citric acid used today.
In one preferred process, the citric acid containing medium is contacted with a solid phase free base resin to absorb the citric acid.
The citric acid is then desorbed by displacement with a stronger acid, and a citric acid containing fraction is collected substantially free from contamination by the stronger acid.
In another preferred process, a citric acid containing medium is contacted with a solid phase divinylbenzene cross-linked polymer to adsorb citric acid, the contacting being at a temperature below about 40 °C. Adsorbed citric acid is then desorbed with hot water.
A typical citric acid process starts with a 15% citric acid broth from a fermentation process and makes a final product of 25% - 30% citric acid feed to customer's evaporator and then crystallizer for final solids processing.
The process steps specified below assumes a fermentation broth feed containing 15% citric acid and some small amounts of salts.
Deashing
The deashing step in the process removes the unwanted salts from the broth by applying ion exchange. The Cations are removed by using a cation resin, then the acid anions which are stronger than citric acid are removed with an anion resin. The anions removed include oxalic, sulfuric and hydrchloric acids.
The broth liquor passes through the Ion-Ix adsorption zone at ambient temperature where the citric acid is adsorbed onto the resin. The spent broth with citric acid removed is send to waste.
The citric acid on the resin is eluted from the resin using hot condensate. The ION-IX system produces a 25 - 30% citric acid solution.
The pretreatment equipment maybe either fixed bed or continuous countercurrent ion exchange ( like ION-IX ).
The resins are regenerated with Na2OH and H2SO4.
A waste stream of NaSO4 is produced.
Lactic Acid process
This lactic process is a simple anion exchange technique on a lactate salt feed broth. Lactic Acid produced via fermentation is extracted typically from an ammonium lactate liquor. The commercial production of purified lactic acid from fermentation broth is usually a process that employs direct acidification followed by ion exclusion, or cation exchange followed by ion exclusion or electrodialysis.
The process employing our ION-IX continuous ion exchange system utilizes a strong(or weak) base resin that cycles through the following process steps: